Prime Minister Narendra Modi on Friday held a meeting to review the country’s vaccination strategy in which important issues related to progress of COVID-19 vaccine development, regulatory approvals and procurement were discussed. In tweets, the Prime Minister said he also reviewed various issues like prioritisation of population groups, reaching out to healthcare workers, cold-chain infrastructure augmentation and technological platform for vaccine rollout.
“Held a meeting to review India’s vaccination strategy and the way forward. Important issues related to progress of vaccine development, regulatory approvals and procurement were discussed,” he said. “Reviewed various issues like prioritisation of population groups, reaching out to HCWs, cold-chain Infrastructure augmentation, adding vaccinators and tech platform for vaccine roll-out,” he added.
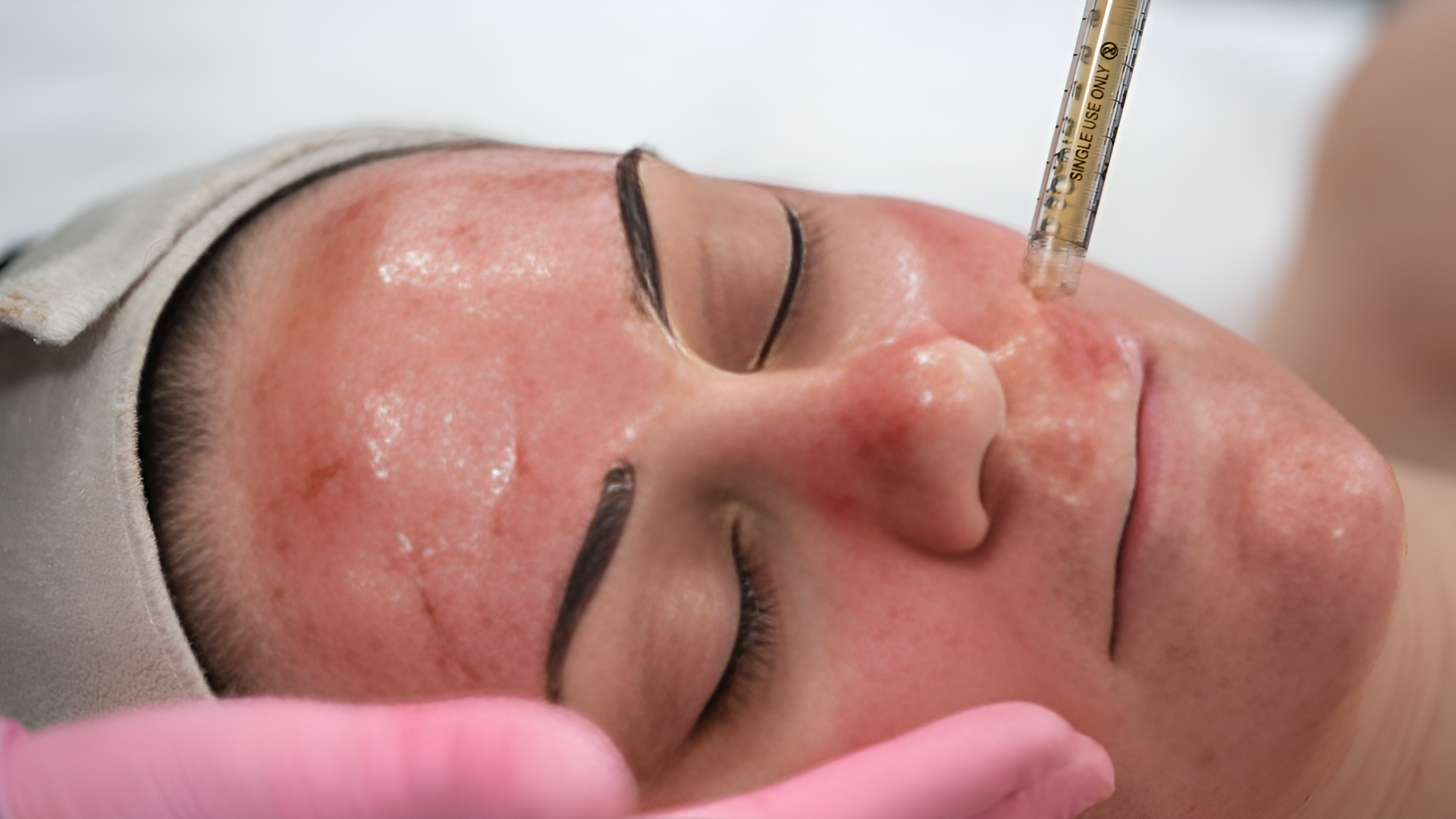
Haryana Health Minister Anil Vij was administered a trial dose of Bharat Biotech’s COVID-19 vaccine ‘Covaxin’ at a hospital in Ambala on Friday. He had offered to be the first volunteer for the third phase trial of Covaxin, which started in the state today.
Also Read: PM Modi chairs meeting on Nagrota Encounter; ‘major attack’ on 26/11 anniversary thwarted: Sources
“It is a matter of pride for India that a company of India has developed a vaccine for coronavirus. Two successful trials have been done and the third phase has begun today. I offer to be the first volunteer so that people without any fear can come forward for vaccination trials. If everything goes well, this vaccine will be in the market soon,” Vij told ANI.
He had offered to be the first volunteer for the third phase trial of Covaxin, which started in the state today. “I will be administered trial dose of Coronavirus vaccine #Covaxin a Bharat Biotech product tomorrow at 11 am at Civil Hospital, Ambala Cantt under the expert supervision of a team of Doctors from PGI Rohtak and Health Department. I have volunteered to take the trial dose,” Vij tweeted on Thursday.
In October, the Drugs Controller General of India (DCGI) gave permission to Bharat Biotech for conducting Phase-III clinical trial for Covaxin. “The DCGI thoroughly analyzed recommendation of the Subject Expert Committee for Bharat Biotech COVID-19 vaccine. On Friday, the top drug regulator granted approval to the pharma giant to conduct phase III clinical trial in India for Covaxin,” a government official had said.
According to the Subject Expert Committee (SEC), on October 5, the firm presented their data from the phase I and II along with animal challenge data in two species including NHP on the Inactivated Corona Virus Vaccine (BBV152) along with the proposal to conduct event-driven phase III clinical trial to assess the efficacy of the vaccine.
Bharat Biotech has partnered with the Indian Council of Medical Research (ICMR) for a country-made COVID-19 vaccine using the virus strain isolated by ICMR’s National Institute of Virology (NIV) based in Pune. The Phase III trials will involve 26,000 volunteers across India, the company said.
Trial volunteers will receive two intramuscular injections approximately 28 days apart. Participants will be randomly assigned to receive COVAXIN™ or placebo. The trial is double-blinded, such that the investigators, the participants and the company will not be aware of who is assigned to which group. COVAXIN has been evaluated in 1,000 subjects in Phase I and Phase II clinical trials, with “promising safety and immunogencity data”.
Also Read: Amid Covid-19 cases surge, Delhi Govt to step up Contact tracing, house-to-house surveys